The long-anticipated new Wegovy pill has just been launched, and here’s everything you need to know about it so far.
Last month (22 December), the Food and Drug Administration (FDA) announced it had approved a pill version of Wegovy (the brand name for semaglutide, a type of GLP-1, from Novo Nordisk), which the NHS can prescribe for weight loss.
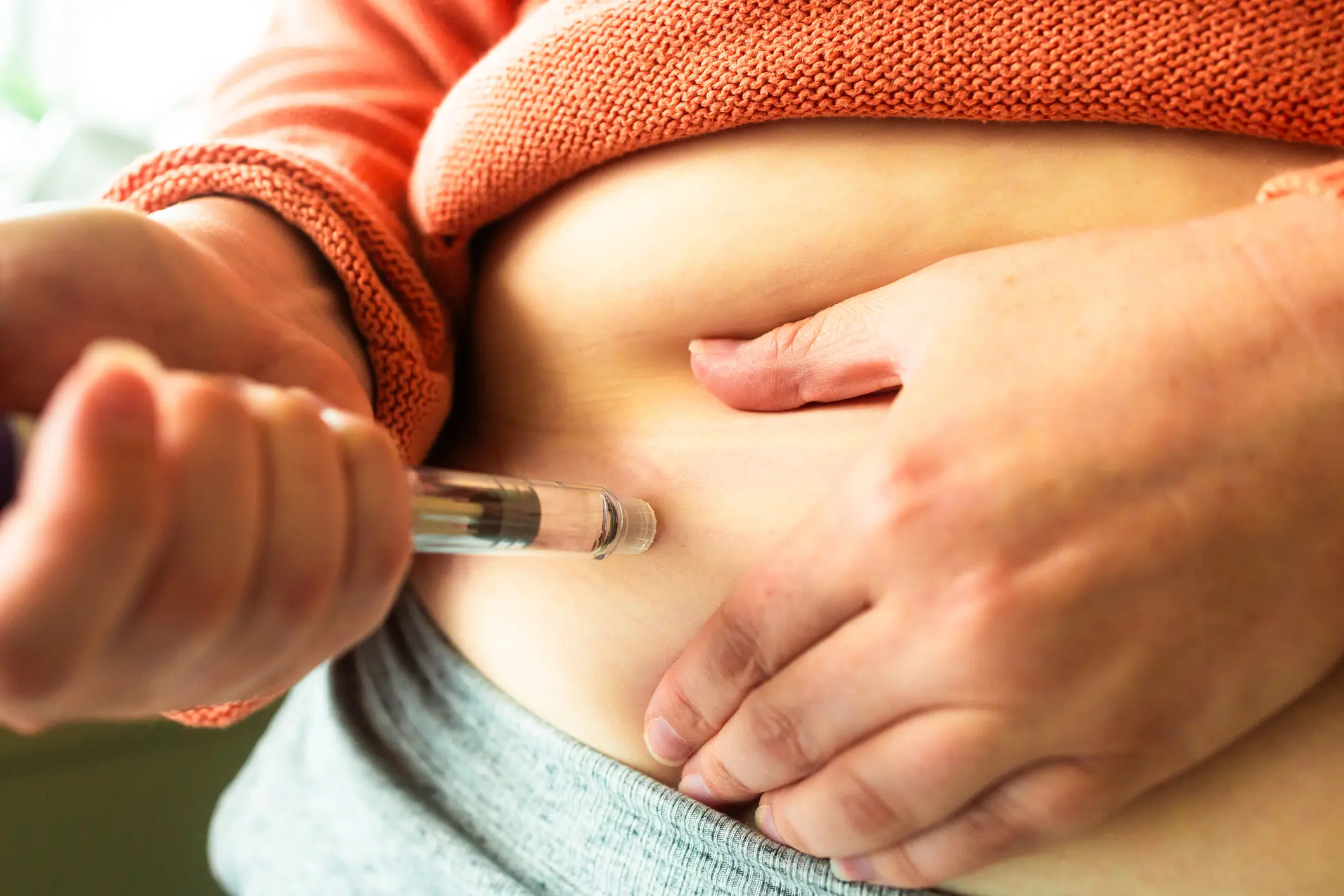
Now, as we know, the current version of many popular weight loss drugs, including Wegovy, requires regular injection, typically administered weekly, but the new version is a once-daily pill that can be taken orally, with no food or drink restrictions.
The pill is currently the first oral version of a GLP-1 drug, though it is expected to be joined by a similar pill from Eli Lilly, which is the manufacturer of Mounjaro.
Fast-forward to now, and the pill is widely available in the United States.
The new Wegovy pill for weight loss has just been launched (Steve Christo - Corbis/Corbis via Getty Images) What is Wegovy?
Wegovy works by mimicking the natural gut hormone glucagon-like peptide-1 (GLP-1), which targets brain regions that regulate appetite and food intake.
This mechanism helps you feel fuller and less hungry, reducing cravings and leading to lower calorie intake and weight loss when combined with diet and exercise.
It's important to note that Novo Nordisk, the manufacturer of Ozempic and Wegovy, has stated that Ozempic is primarily intended for adults with type 2 diabetes, rather than for those seeking to lose weight.
Meanwhile, Wegovy, which is also semaglutide, is FDA-approved for weight-loss management.
Wegovy works by mimicking a natural gut hormone, glucagon-like peptide-1 (GLP-1) (coldsnowstorm / Getty Images) How will the Wegovy pill be different from the Wegovy injection?
Well, for starters, it will be taken orally as opposed to injected into the body.
"This is a meaningful step forward in the field," Dr Christopher McGowan, a gastroenterologist who runs a weight loss clinic in Cary, North Carolina, told NBC News. "It won’t replace injectables, but it broadens our tool kit in an important way."
"Pills are familiar, nonintimidating and fit more naturally into most people’s routines," he added. "For many patients, a pill isn’t just easier, it’s psychologically more acceptable."
However, the pill form still requires a prescription from a doctor.
"Oral GLP-1s such as Wegovy pill represent a real breakthrough, and help address long-standing barriers like access, friction, and affordability," Scott Honken, Chief Commercial Officer at Weight Watchers, told PEOPLE, calling the Wegovy pill a 'more efficient and impactful treatment'.
In terms of possible side effects, McGowan said they may feel 'more intense' as the medication will be digested by the stomach, which could trigger nausea.
Experts have hailed the Wegovy pill launch as a 'real breakthrough' (WLADIMIR BULGAR/SCIENCE PHOTO LIBRARY / Getty Images) How effective are the Wegovy pills?
Trials have shown the pills to be as effective as the injections when taken alongside a reduced-calorie diet and exercise.
Results from a Phase 3 trial published in the New England Journal of Medicine showed that participants taking the highest dose of the Wegovy pill lost an average of 16.6 per cent of their body weight over 64 weeks, compared with 2.2 per cent in the placebo group.
That aligns closely with the injectable Wegovy, which produced about 15 per cent weight reduction after 68 weeks.
"This moment is about changing what's possible in weight management, and to make that possible, we have worked to ensure Wegovy pill is affordable and accessible to those who need it, however they choose to receive their care," Ed Cinca, Novo Nordisk’s senior vice president, Marketing & Patient Solutions, said in a statement from the company.
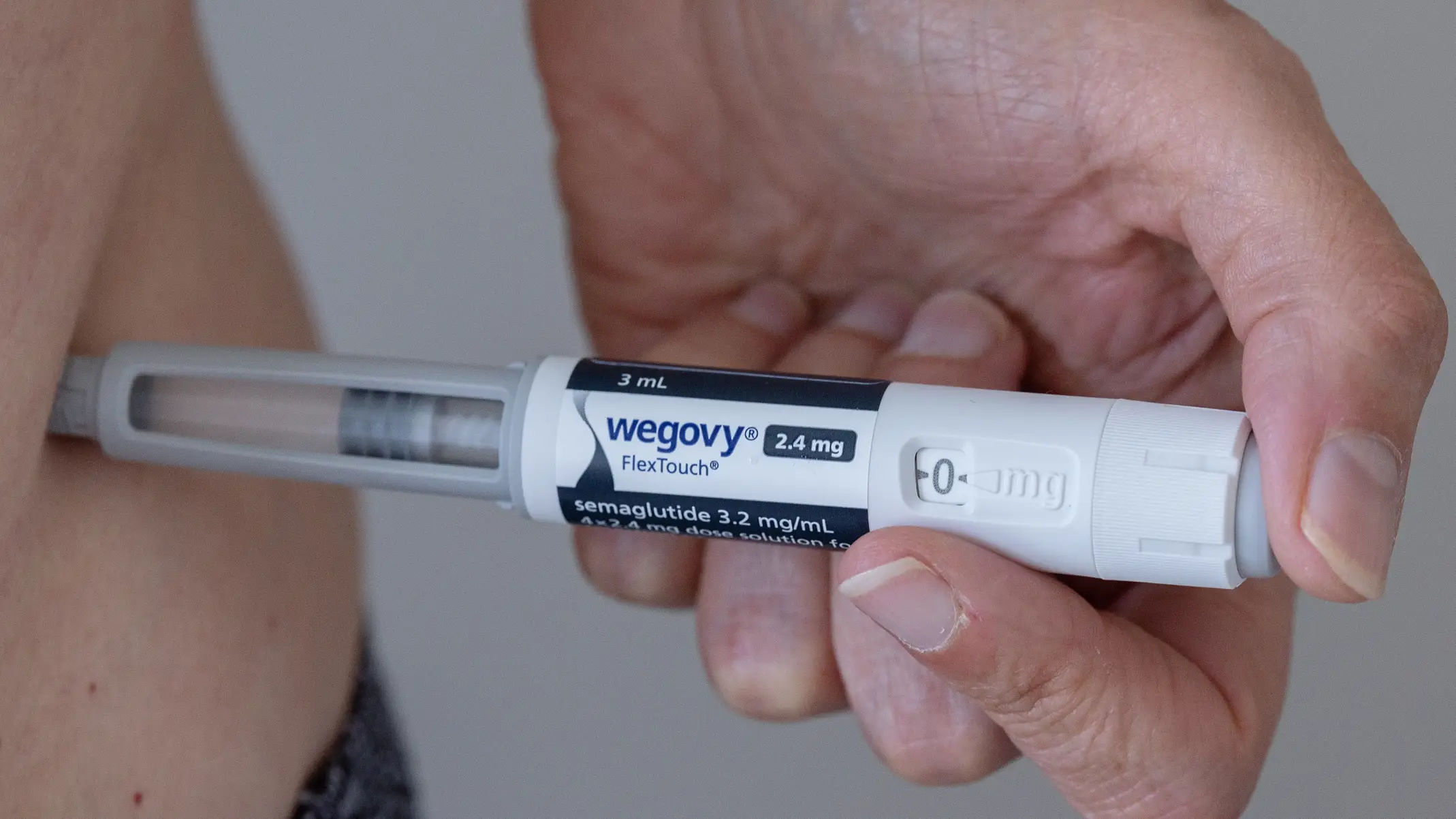
The Wegovy pill can cost as little as $25 a month (Grace Cary / Getty Images) How much will the Wegovy pill cost?
The Wegovy pill can cost as little as $25 a month for those whose insurance covers obesity medication.
Novo Nordisk has explained that there is also a self-pay option, which works out to be around $5 a day, or $149 a month, for the starting dose of 1.5 mg.
A discounted price of the next dose, 4 mg, will cost $149 a month until 15 April, after which it’ll increase to $199 a month.
Meanwhile, higher doses (9 mg and 25 mg) will cost $299 per month.
Similar to the injections, the pill will come in several doses, with patients typically starting at the lowest dose before gradually increasing over several weeks to adjust to any side effects.
As for whether it will be available in the UK, according to The Guardian, the UK's medicine regulator is reviewing Novo Nordisk's application and is expected to reach a decision by the end of the year.
 Rhiannon Ingle
Rhiannon Ingle