Life
The latest news, health, travel and lifestyle stories from around the world, all in one place.
The latest news, health, travel and lifestyle stories from around the world, all in one place.
The latest news, health, travel and lifestyle stories from around the world, all in one place.

The new findings were presented at the European Congress on Obesity

Social media users have opened up about the 'primal' kink

When was the last time you gave your electric toothbrush a proper once over?

We're just a few days away from the major cosmic event lighting up our skies

A simple 12-second sing-along was the social media favourite's calling card for fame
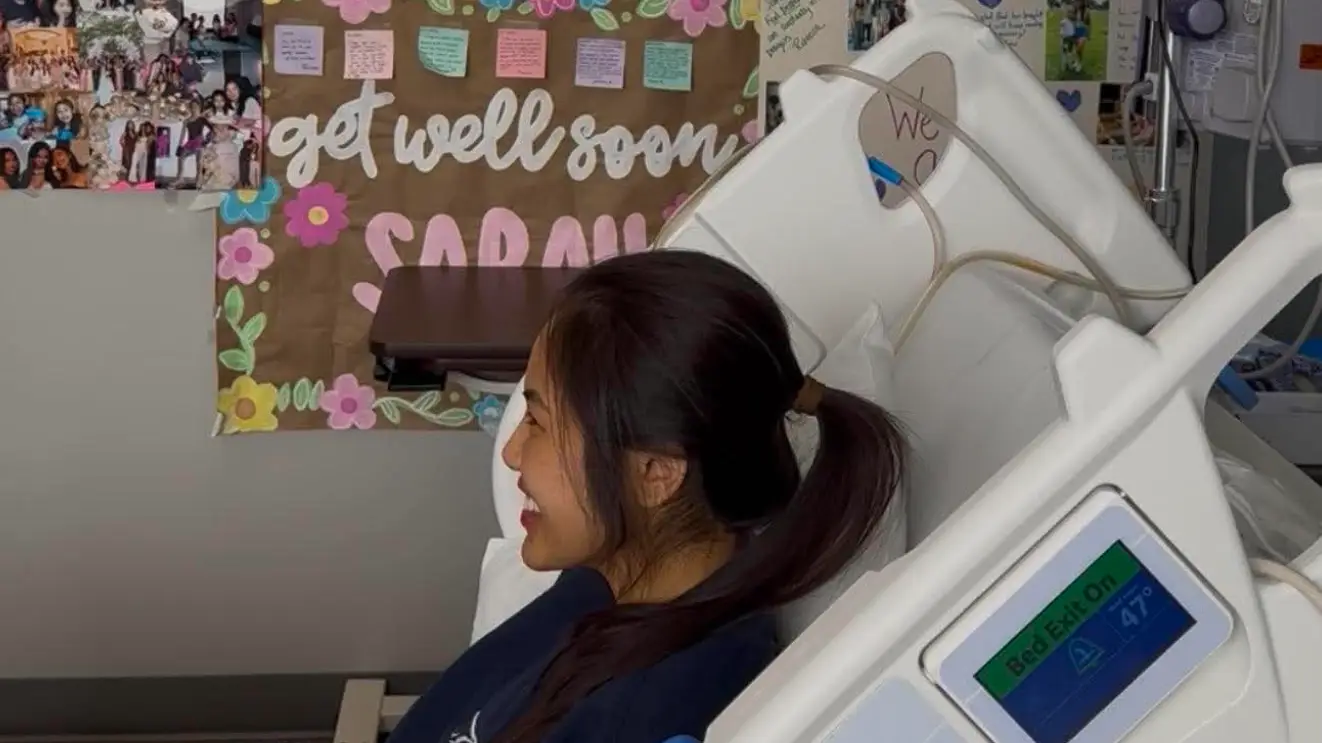
US nurse Sarah Danh, 27, has been fighting for her life after she unexpectedly suffered acute liver failure on day two of her honeymoon

Nicky Wake spoke to Tyla about her experience with a phenomenon known as 'widow's fire'

Whoopi Goldberg, real name Caryn Johnson, revealed why her mother pushed her to scrap her original stage name

Dr Francesco Lo Monaco has revealed the six things that should be avoided in the evening
You may have heard of 'nutmaxxing', but 'ballmaxxing' is a whole other kettle of fish...

The National Institute for Health and Care Excellence (NICE) has revealed that doctors now know blue inhalers can 'make the condition worse'

The X-ray of a car passenger who had her feet on the dashboard during a crash was shared by police as a warning against the dangerous move

Psychologists have explained the real reason we cry on our birthdays - and it's all to do with 'zenosyne'

The full Flower Moon in Scorpio is set to rise tonight - and an astrologer has warned that four star signs will feel its impact the most

Jonah Hill made the surprising move to relocate away from Hollywood and has now confessed why

Who knew there was a right and wrong way for a number two

Hospice nurse and educator, Katie Duncan, shared the one common thing she noticed among patients in end-of-life care

Experts have outlined a number of baby names from the 1940s, 50s, and 60s that are rising in popularity in 2026

Dr Jeremy London outlined the four things he 'absolutely avoids as a heart surgeon'

Channel 4's Virgin Island season two kicked off this week with a brand new batch of people hoping to overcome their struggles with intimacy

Virgin Island contestant Joy, 22, said that at one point, she heartbreakingly believed 'God cursed her' with vaginismus

The Jersey Shore icon has opened up two months after revealing she had the disease

The everyday painkiller is thought to prevent certain tumours from forming and spreading across the body

A doctor has revealed what could be 'reducing the effectiveness' of your antihistamines as the pollen count is on the rise in the UK