A popular eczema cream has now been recalled after potentially life-threatening bacteria were found to have contaminated certain lots.
The US Food and Drug Administration (FDA) released an announcement last week (8 May) titled: "Pharmacal Issues Nationwide Recall of MG217 Multi-Symptom Treatment Cream & Skin Protectant Eczema Cream Due to Microbial Contamination."
This is because the product was found to be contaminated with Staphylococcus aureus.
The NHS explains that staph infections are caused by bacteria called Staphylococcus, and they most often affect the skin.
Symptoms of a staph skin infection can include: a painful red lump or bump (a boil, carbuncle or abscess), hot, red and swollen skin (cellulitis), sores, crusts or blisters (impetigo) and sore, red eyelids or eyes (styes or conjunctivitis).
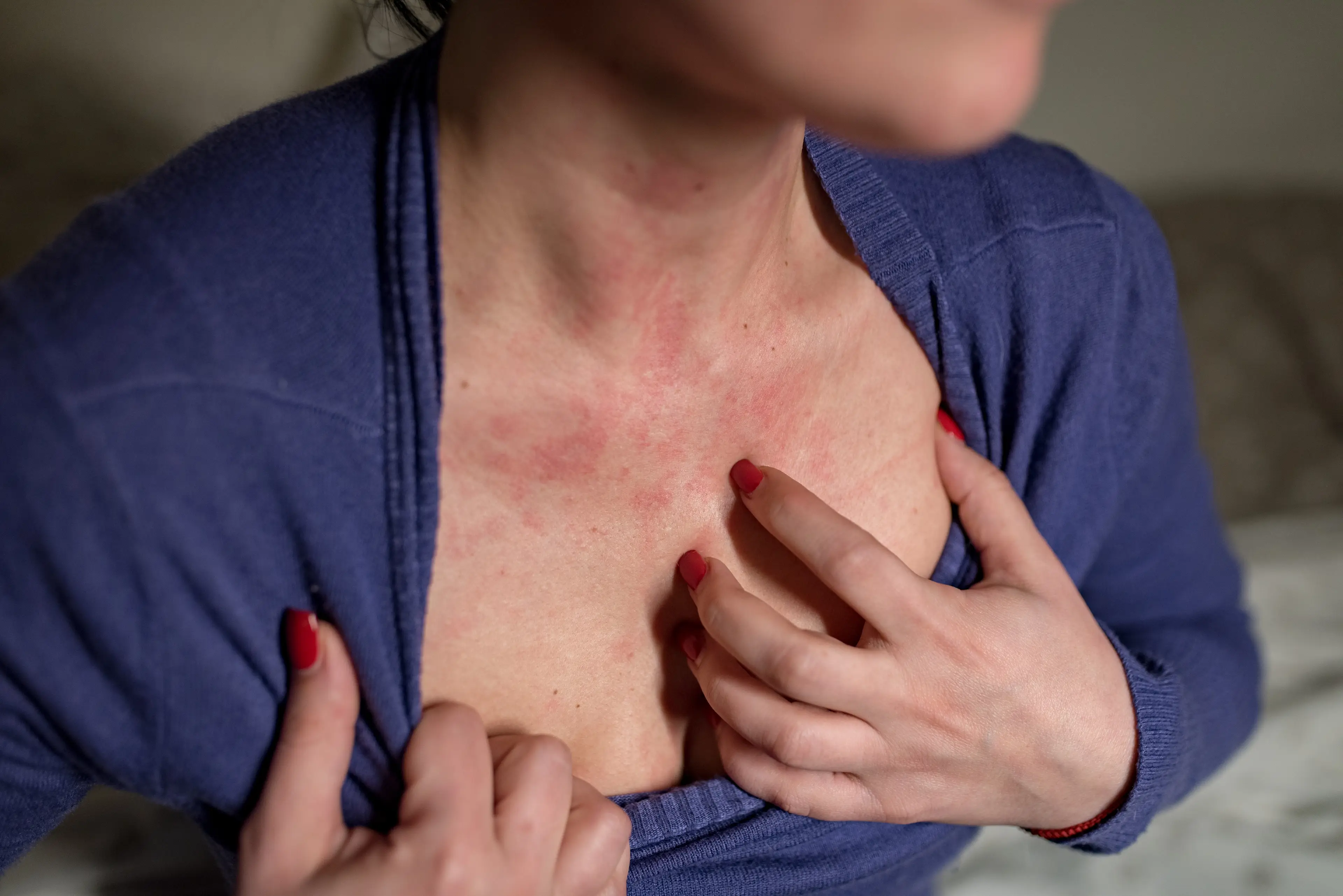
The FDA has warned that use of the recalled product could result in 'life-threatening adverse events' (Getty Stock Images) The bacteria that cause staph infections live harmlessly on many people's skin, often in the nose, armpits and on the buttocks.
They usually only cause an infection if they get into the skin, such as through a bite or cut.
Staph bacteria can spread to others through close skin contact, sharing things like towels or toothbrushes and droplets in coughs and sneezes (but this is less common).
The health body warns that Staph bacteria can also cause more serious infections, like blood poisoning and toxic shock syndrome. However, these are much less common than skin infections.
The FDA's Risk Statement reads: "Use of the product could result in a range of infections from localised to severe or life-threatening adverse events.
"Patients with weakened immune systems or compromised skin — such as those with wounds, burns, or skin disorders — are at increased risk for serious infections, including skin and skin structure infections, infective endocarditis (infection of the heart valves), bone and joint infections, bloodstream infections, and life-threatening conditions like sepsis and septic shock.
"To date, Pharmacal has not received any reports of adverse events related to this recall."
The affected MG217 Multi-symptom Treatment Cream & Skin Protectant Eczema Cream is lot 1024088 with expiration date of November 2026 (FDA) The official press release outlines that Pharmacal is recalling one lot of MG217 Multi-symptom Treatment Cream & Skin Protectant Eczema Cream, 6oz tube, to the consumer level.
The topical cream's product code is 5106 and UPC 012277051067.
The affected MG217 Multi-symptom Treatment Cream & Skin Protectant Eczema Cream is lot 1024088 with an expiration date of November 2026.
As per the FDA, Pharmacal is notifying its distributors via an emailed letter and is arranging for the return of all recalled products. Consumers who have a product which is being recalled should stop using and discard the product.
Tyla has reached out to Pharmacal for further comment.
 Rhiannon Ingle
Rhiannon Ingle